
Applications
At WhiteLab Genomics, we empower our partners to optimize genomic medicine development through in silico methods that reduce costs and accelerate timelines. Our AI-driven approach accelerates the development of breakthrough solutions in genomic medicine, saving resources while delivering precise, effective therapies.
Target Receptor Identification
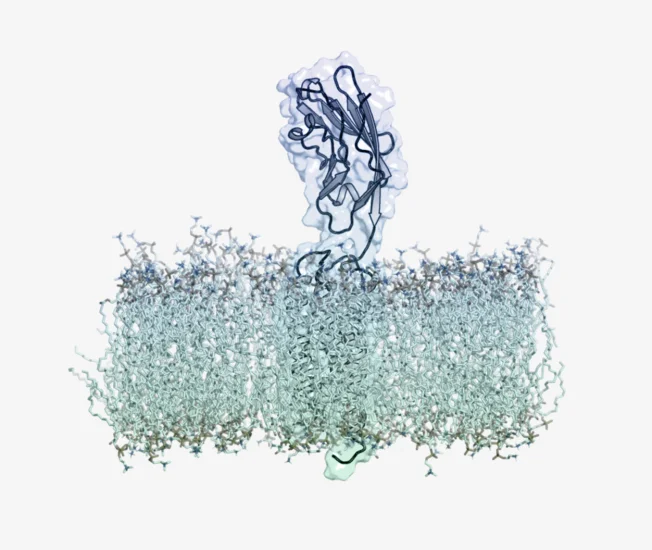
Biomarker identification: Identify markers that are specific to your target tissue/cell type
Receptor validation: Optimise choice of animal models and avoid targets that present biological risks
Powered by Protein Language Models & Deep Learning

Vector Engineering
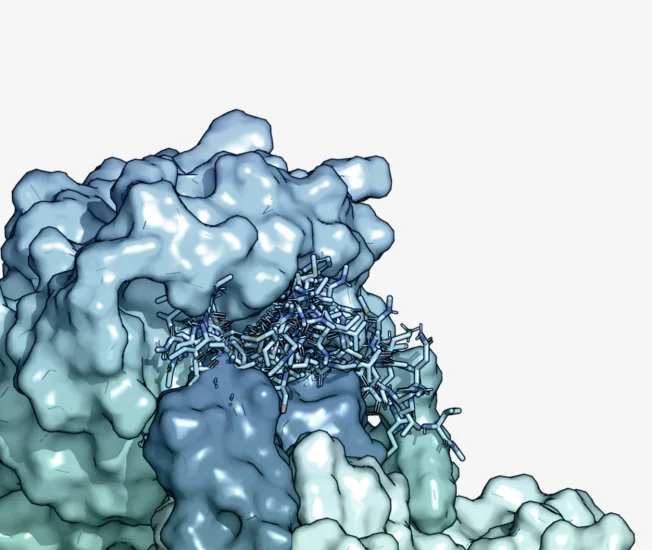
Advanced Peptide Engineering: Generate high-affinity vectors for targeted delivery
In Silico Optimization: Design vectors with data & physics driven methods for structural biology analysis
Enabled by Generative AI, Machine Learning & Physics based Algorithms
Payload Design
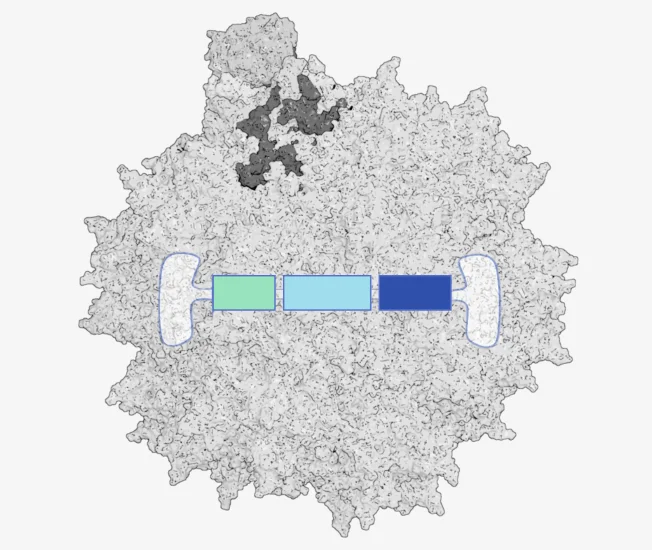
- Functional Modulation: Adjust activity and tissue specificity of therapeutic molecules
- Minimize Side Effects: Ensure efficacy while reducing adverse effects
Powered by Generative AI

Cell Therapy
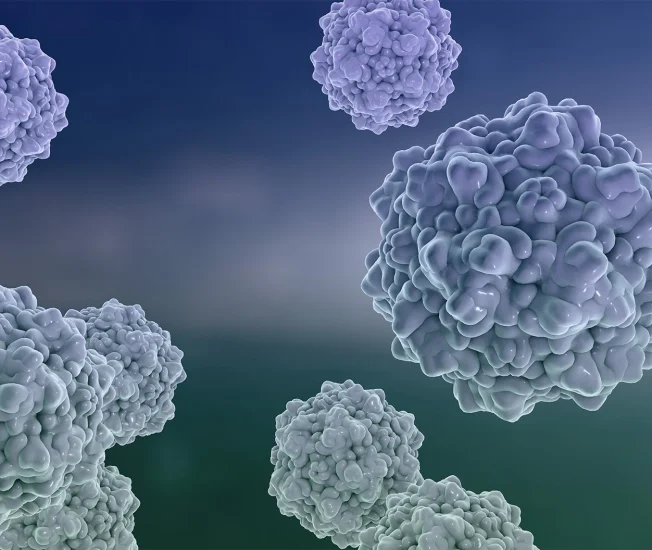
- Antigen Targeting: Select appropriate antigens and binding sites
- On-target activity: Design CARs with improved on-target activity and reduced cytotoxicity
Bioproduction
- Conducive characteristics: Identify traits for high vector production yield
- Optimize factors: Improve gene expression, metabolic activity, and growth rates
Guided by Biostatistics
